MAJOR Docusate Sodium 250 mg, Extra Strength Stool Softener Laxative for Occasional Constipation Relief – Stimulant‑Free, 100 Softgels (1 Pack)
Stock Expiry Date: 09-02-2027
Made in the USA
100% Authentic — Sourced directly from manufacturers or their authorized dealers!
- STOOL SOFTENER LAXATIVE – Docusate Sodium 250 mg softgels help ease occasional constipation by gently softening dry, hard stools, making bowel movements more comfortable and natural within 12 to 72 hours.
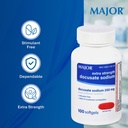
- SINGLE-ACTION, STIMULANT-FREE SUPPORT – These stool softeners work without harsh stimulant laxatives, providing a gentle, non-habit-forming option for occasional constipation relief.
- HOW TO USE - Adults and kids 12+: take 1 docusate sodium 250mg softgels daily. Taken preferably at bedtime or as directed by a doctor.
- QUALITY, EXTRA STRENGTH FORMULATION – Formulated with 250mg of docusate sodium, these softgels are designed for those seeking a dependable, stimulant-free, extra strength laxative option, safe for daily use.
- MAJOR PHARMACEUTICALS – At Major Pharmaceuticals, we believe wellness should be accessible to everyone. We pride ourselves on our legacy of delivering affordable, high-quality OTC and vitamin supplements, trusted by healthcare professionals and pharmacies across 50+ therapeutic categories.
| Product Benefits | Constipation Control, Stool Softener |
| Ingredients | Sorbitol, Purified Water, Fd&c Red #40, FD&C Blue #1, FD&C Yellow #6, sorbitan, Gelatin Glycerin, Docusate Calcium 240 mg - Stool softener laxative, Inactive Ingredients: Corn Oil |
| Special Ingredients | Latex Free, Gluten Free, Soy Free, Peanut Free, Preservative Free |
| Allergen Information | Soy Free, Peanut Free, Gluten Free, Preservative-Free |
| Net Content Count | 1 Bottle, 100 Count |
| Brand | Major |
| Flavor | Unflavored |
| Unit Count | 100 Count |
| Item Form | Softgel |
| Item Weight | 4.16 Ounces |
| Number Of Items | 1 |
| Dosage Form | Softgels |
| Manufacturer | Major Pharmaceuticals |
| Product Type | Laxatives |
| Specific Uses For Product | Constipation |
| Active Ingredients | Docusate Sodium 250mg |
| Upc | 305363755018 |
| Package Type Name | Box |
| Target Audience | Unisex-Adults |
| Brand Name | MAJOR |
| Age Range Description | Adult |
| Container Type | Bottle |
| Manufacturer Part Number | 305361065010-1-A1 |
| Item Dimensions | 2.06 x 3.88 x 2.06 inches |
-
5% off when you buy 4 or more.
-
Get 5% off your next purchase.
-
Orders are processed and shipped from Brooklyn, USA.
-
Enjoy free delivery on orders above AR$ 290,000.
3-6 days (Domestic)
30-Day Return Guarantee
Confidence-backed purchases with 30-day return support.
SKU |
BRSWIYIBOYOAGDQ3 |
Category |
Health Care , Over-the-Counter Medication , Digestion & Nausea , Laxatives |
Barcode |
305363755018 |
Tags |
Important information
Ingredients
Docusate Calcium 240 mg - Stool softener laxative, Inactive Ingredients: Corn Oil, FD&C Blue #1, FD&C Red #40, FD&C yellow #6, Gelatin Glycerin, Purified Water, Sorbitan, Sorbitol
Legal Disclaimer
Statements regarding dietary supplements have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease or health condition.
Product Dimensions
2.06 x 3.88 x 2.06 inches; 4.16 ounces
Shipping & Delivery
At Nutricity, we aim to deliver your order as quickly and safely as possible.
Domestic Shipping :
Orders are typically delivered within 3–6 business days.
International Shipping:
Estimated delivery time is 12–26 business days, depending on the destination and customs processing.
You will receive a tracking number once your order has been dispatched. Delivery times may vary during holidays or due to unforeseen delays.
Disclaimer
Dietary supplements are not medicines. Products sold on this website are not intended to diagnose, treat, cure, or prevent any disease. If you have a medical condition then always consult a qualified healthcare professional before use. Individual results may vary. By placing an order, you agree to comply with your country’s import regulations. We cover applicable duties and taxes where stated.